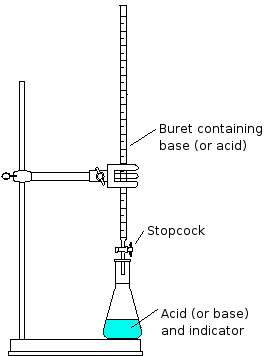
Titrations
A titration is a process of neutralizing that slowly adds one solution (titrant) to another solution until it reaches neutralization.
To help explain this process, here's an example:
|
Let's say you are trying to neutralize NaOH with HCl. First, you would put 10 cc of NaOH in a flask. Next, using a burette, you would try and drip as close to 10 cc of HCl into the flask, so that the neutralization of NaOH and HCl can be exact. Preciseness is key in a titration because adding one extra drop can change a neutral solution to something acidic or basic.
For extra help, click the button below to watch a video on the process of titration. |
Click here to look at a specific titration experiment procedure with NaOH and HCl.