Making Salts
Salts are produced by mixing an acid & a base, an acid & an alkali, an acid & carbonate, an acid & metal (as spoken about in previous).
Chlorides, sulfates, nitrates are large group of salt compounds. The compound that is produced is dependent on the acids you use, since it determines the family of the salt, while the base determines what specific type of the salt family is produced. Here are examples of salt compounds produced using three strong acids:
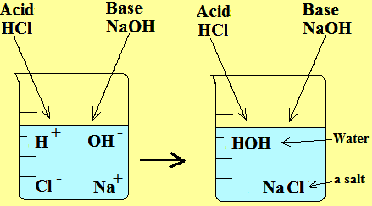
1. Hydrochloric acid (HCl) + Sodium hydroxide (NaOH) --- NaCl (sodium chloride) + H2O (water)
1. Hydrochloric acid (HCl) + Sodium hydroxide (NaOH) --- NaCl (sodium chloride) + H2O (water)
2. Sulfuric acid (H2SO4) + Potassium hydroxide (KOH) --- K2SO4 (potassium sulfate) + H2O
(water)
3. Nitrate Acid (HNO3) + Lithium hydroxide (LiOH) --- LiNO3 (Lithium nitrate) + H2O (water) Salt is formed is a result of neutralization of acids and bases
(water)
3. Nitrate Acid (HNO3) + Lithium hydroxide (LiOH) --- LiNO3 (Lithium nitrate) + H2O (water) Salt is formed is a result of neutralization of acids and bases