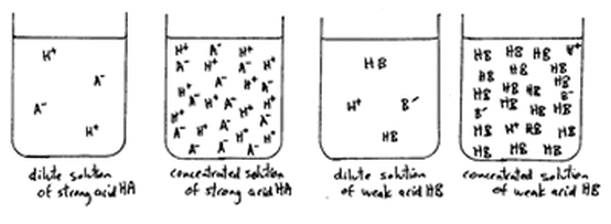
Concentration vs. Strength
Concentration of a liquid is the amount of the acid/base per unit volume (liter), while the strength of a liquid is the ability of the acid/base to loose hydrogen ions/hydroxide ions. The connection between concentration and strength is that strength is dependent on the concentration of hydrogen ions produced (for acid)/hydroxide ions produced (for base).
A concentrated solution is one that has a large amount of acid or base per unit volume. A diluted solution is one that has a small amount of acid or base per unit volume.
A concentrated solution is one that has a large amount of acid or base per unit volume. A diluted solution is one that has a small amount of acid or base per unit volume.
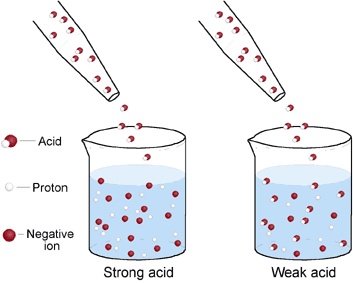
A strong acid is one that dissociates completely in a solution to form hydrogen ions. A strong base is one that dissociates completely in a solution to form hydroxide ions. Conversely, a weak acid dissociates partially or not at all in a solution to form hydrogen ion. A weak base dissociates partially or not at all in a solution to form hydroxide ions.
Click here to learn about conjugate acids and conjugate bases.