Neutralization rEACTION of Acids and bases
|
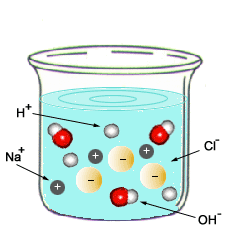
When mixing an acid and a base, both solutions become neutral. This means that the pH level of a neutralization reaction is equal to 7. During the neutralization of acids and bases, the reaction produces salt and water.
Acid + Base --- Salt + Water Example:
|
nEUTRALIZATION OF aCIDS
Acids can be neutralized four different ways. In all methods of acid neutralization, it produces salt.
- Acid + base ---> salt + water
- Acid + alkali ---> salt + water
- Acid + metal ---> salt + hydrogen gas (H2)
- Acid + carbonate (CO3)--- > salt + water + carbon dioxide (CO2)
Click here to learn about how mixing acids and bases can make salts.