BUFFERS
In experiments, sometimes it is necessary to have a constant pH when mixing solutions - this is the purpose of buffers. Buffers can resist a change in pH even when acidic or basic solutions are added. Buffers work by neutralizing these acidic or basic components and keeping the pH stable. Explicitly, buffers keep an equal amount of conjugate base and conjugate acid present so it's able to neutralize other amounts of H+ or OH- added into a solution.
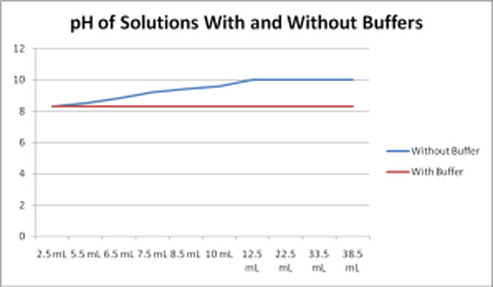
Here's a diagram that outlines the effect of a buffer on pH:
Here's a diagram that outlines the effect of a buffer on pH:
A buffer is composed of a weak conjugate acid-base pair. This means that either a buffer will consist of either a weak acid and its conjugate base or a weak base and its conjugate acid. Choosing whether to use a weak base or weak acid depends on what the desired pH level of the solution is.
|
Acidic Buffer Example:
CH3COOH + NaOH ---> CH3COONa + H2O |
Basic Buffer Example:
NH3 + HCL --> NH4CL (Explanation - When a HCl is added with NH3, it binds together the H+ with the ammonium ion, thus the proton cannot affect the pH level.) |
Click here to learn about real life uses of buffers.