General INFO
Definitions:
- Acid – chemical species that donates protons or hydrogen ions
- Base – chemical species that donates electrons or hydroxide ions
- Acid = mixing a non-metal oxide and water
- Examples of Acids: HCl, H2SO4, HNO3
- Base = mixing a metal oxide and water
- Examples of Bases: NaOH, NH4OH, NH3
|
Properties of Acids:
|
Properties of Bases:
|
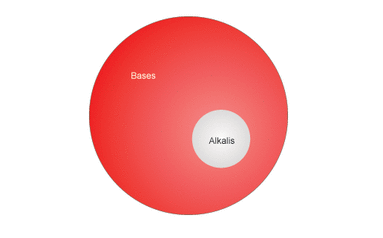
Bases vs. Alkalis
- Alkalis – bases that dissolve in water: Examples - NaOH (Sodium Hydroxide), NH3(Ammonia) and KOH (Potassium Hydroxide).
- Bases – solutions that don’t dissolve in water
Click here to continue and learn about acid and base theories.