pH Scale
pH measures the hydrogen ion concentration in a liquid, which is the strength of a liquid. pH stands for "power of hydrogen". The pH scale is separated in three catagories: acids, neutral liquids, and bases. Acids are found between any number lower than exactly 7 and 1, while bases can be found between any number higher than exactly 7 and 14. Solutions that have a pH level of 7 are neither acidic or basic, they are neutral. An example of a neutral liquid is distilled water.
*FYI: The liquids you usually find in every day life have a pH level close to 7.
Closer look at the pH scale:
*FYI: The liquids you usually find in every day life have a pH level close to 7.
Closer look at the pH scale:
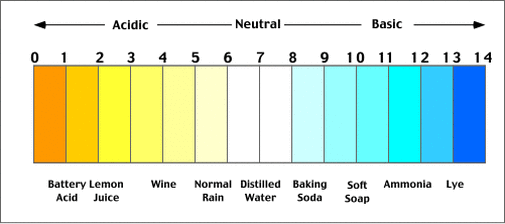
- 1-3 is a strong acid
- 4-6 is a weak acid
- 7 is neutral
- # larger than 7-10 is a weak base
- 11-14 is a strong base
pH can also help you find out [H+], [OH-], and pOH in a liquid. Finding [H+] is calculating the amount of protons in a solution, and finding [OH-] is calculating the amount of hydroxide ions in a solution. Finding pOH is calculating the "power of hydroxide ions".
1. Relationship between pH and H+
pH = -log[H+]
[H+] = 2nd function log [-pH]
2. Relationship between pH and pOH
pH + pOH = 14
3. Relationship between pOH and OH-
pOH = -log[OH-]
[OH-] = 2nd function log[-pOH]
NOTE: 2nd function log = 10^(___)
Example: Find [OH-]
2nd function log [-pOH] = 10^(-pOH)
1. Relationship between pH and H+
pH = -log[H+]
[H+] = 2nd function log [-pH]
2. Relationship between pH and pOH
pH + pOH = 14
3. Relationship between pOH and OH-
pOH = -log[OH-]
[OH-] = 2nd function log[-pOH]
NOTE: 2nd function log = 10^(___)
Example: Find [OH-]
2nd function log [-pOH] = 10^(-pOH)
|
Practice Question 1:
pH = 5.36 Find [H+], [OH-], and pOH. Calculations: pOH = 14 - 5.36 = 8.64 [OH-] = 2nd function log [-8.64] = 2.29 x 10^-9 [H+] = 2nd function log [-5.36] = 4.37 x 10^-6 |
Practice Question 2:
[OH-] = 4.07 x 10^-12 Find [H+], pOH, and pH. Calculations: pOH = -log[4.0x x 10^-12] = 11.39 pH = 14 - 11. 39 = 2.61 [H+] = 2nd function log [-2.61] = 2.45 x 10^-3 |
Well, how do you feel about these practice problems?! Confident? Click here to test your skills with a pH quiz.
