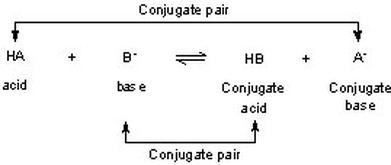
Conjugate acids and Conjugate bases
According to the Bronsted Lowry Theory of acids a bases, a conjugate acids are a substances created after a base receives a proton (H+), while a conjugate base is a substance left over after an acid loses a proton (H+). This proton transfer occurs in an acid-base reaction.
Example:
HNO3 + H2O ---> H3O+ + NO-3
HNO3 is an acid in this situation because it gives a proton to to H2O, yielding, H3O. As a result, HNO3 is left as NO-3.
Because HNO3 is the acid, H2O in this situation must be the base. H3O in this reaction is the conjugate acid since it is the proton acceptor.
Acid Base C-Acid C-Base
HNO3 + H2O ---> H3O+ + NO-3
HNO3 + H2O ---> H3O+ + NO-3
HNO3 is an acid in this situation because it gives a proton to to H2O, yielding, H3O. As a result, HNO3 is left as NO-3.
Because HNO3 is the acid, H2O in this situation must be the base. H3O in this reaction is the conjugate acid since it is the proton acceptor.
Acid Base C-Acid C-Base
HNO3 + H2O ---> H3O+ + NO-3